Anti-Lck (phospho Y504) antibody (ab4901)
Key features and details
- Rabbit polyclonal to Lck (phospho Y504)
- Suitable for: WB
- Reacts with: Recombinant fragment
- Isotype: IgG
Overview
-
Product name
Anti-Lck (phospho Y504) antibody
See all Lck primary antibodies -
Description
Rabbit polyclonal to Lck (phospho Y504) -
Host species
Rabbit -
Specificity
Hck [pY522] (75% homology), and Lyn [pY508] (83.3% homology) have not been tested. -
Tested Applications & Species
See all applications and species dataApplication Species WB Recombinant fragment -
Immunogen
-
Positive control
- Full length untagged recombinant human Lck protein.
-
General notes
Lck (p56lck), a member of the Src family of non-receptor tyrosine protein kinases, is expressed predominantly in T cells. Lck function is critical both for T cell development in the thymus and activation of mature T cells in the periphery by antigen. The activity of Lck is regulated by phosphorylation of two conserved tyrosine residues, Tyr-505 (equivalent to Tyr-529 in c-Src) and Tyr-394 (equivalent to Tyr-418 in c-Src). Tyr-505 is located near the carboxyl terminus of Lck and, when phosphorylated, associates intramolecularly with the SH2 domain in the amino-terminal half of the protein. This helps stabilise Lck in a conformation that, biologically, is relatively inactive. In the absence of phosphorylation at Tyr-505, intramolecular binding of the carboxyl terminus to the SH2 domain does not occur, and Lck exhibits increased activity in vivo.
Properties
-
Form
Liquid -
Storage instructions
Shipped at 4°C. Upon delivery aliquot and store at -20°C or -80°C. Avoid repeated freeze / thaw cycles. -
Storage buffer
pH: 7.3
Preservative: 0.05% Sodium azide
Constituents: PBS, 0.1% BSA
BSA is IgG and protease free -
 Concentration information loading...
Concentration information loading... -
Purity
Immunogen affinity purified -
Purification notes
Purified from rabbit serum by sequential epitope-specific chromatography. The antibody has been negatively preadsorbed using (i) a non-phosphopeptide corresponding to the site of phosphorylation to remove antibody that is reactive with non-phosphorylated Lck, and (ii) a generic tyrosine phosphorylated peptide to remove antibody that is reactive with phospho-tyrosine (irrespective of the sequence). The final product is generated by affinity chromatography using a Lck-derived peptide that is phosphorylated at tyrosine 505. -
Primary antibody notes
Lck (p56lck), a member of the Src family of non-receptor tyrosine protein kinases, is expressed predominantly in T cells. Lck function is critical both for T cell development in the thymus and activation of mature T cells in the periphery by antigen. The activity of Lck is regulated by phosphorylation of two conserved tyrosine residues, Tyr-505 (equivalent to Tyr-529 in c-Src) and Tyr-394 (equivalent to Tyr-418 in c-Src). Tyr-505 is located near the carboxyl terminus of Lck and, when phosphorylated, associates intramolecularly with the SH2 domain in the amino-terminal half of the protein. This helps stabilise Lck in a conformation that, biologically, is relatively inactive. In the absence of phosphorylation at Tyr-505, intramolecular binding of the carboxyl terminus to the SH2 domain does not occur, and Lck exhibits increased activity in vivo. -
Clonality
Polyclonal -
Isotype
IgG -
Research areas
Images
-
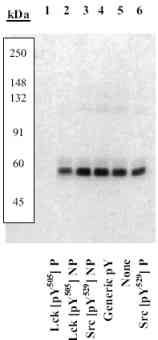
Peptide Competition: Full length untagged recombinant human Lck protein was added to control cell extract which does not contain Lck and then resolved by SDS-PAGE on a 10% Tris-glycine gel. The proteins then were transferred to nitrocellulose and incubated with 0.50
µ g/mL ab4901 antibody, following prior incubation with: (1) the appropriately phosphorylated peptide immunogen, (2) the nonphosphorylated peptide corresponding to the peptide immunogen, (3) the non-phosphorylated peptide derived from the corresponding region of Src, (4) a generic phosphotyrosine containing peptide, (5) no peptide, and (6) the phosphorylated peptide derived from the corresponding region of Src. After washing, membranes were incubated with goat F(ab’)2 anti-rabbit IgG alkaline phosphatase conjugate and bands were detected using the Tropix WesternStar detection method. The data presented here demonstrate that only the phosphopeptide corresponding to this site completely blocks t